Revatis S.A. • Markets & usage • Growth prospects for cell therapy in veterinary medicine
The fields of application of cell therapy are broadly similar to those emerging in human medicine.
Ethics and animal welfare
In 2018, after the FDA, the European agency regulated the use of cell therapy in veterinary medicine by requiring GMP production, proof of safety and efficacy demonstrated by approved clinical studies. Revatis adheres to this quality approach and is now conducting several clinical trials in horses and shortly in companion animals.

The Revatis proposal in regenerative medicine
Revatis offers veterinarians and horse owners a full range of products and services aimed at improving the well-being and performance of their clients’ animals. Regenerative medicine offers innovative and effective treatments for tendon and joint pathologies in sport and leisure horses.
Kit PRP Tendon Axovet
Medical device for the preparation of autologous platelet-rich plasma.
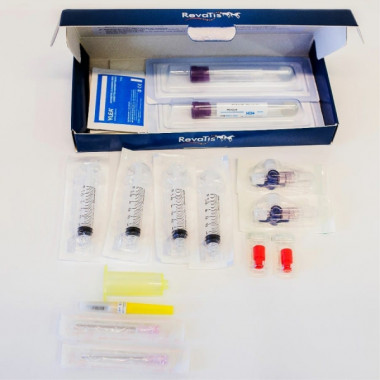
Kit PRP Joint Axovet
Medical device for the preparation of autologous platelet-rich plasma associated with hyaluronic acid
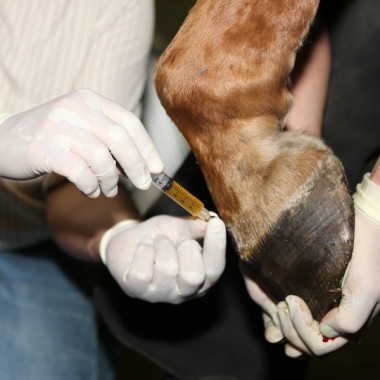
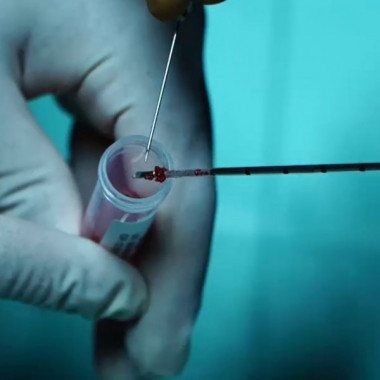
Autologous muscular derived stem cells
Sampling kit
The development of innovative cellular products for therapeutic purposes
Revatis contributes to the development of cell therapy as a validated treatment for traumatic, inflammatory, degenerative and autoimmune pathologies in horses and companion animals. New perspectives are opening in the biotechnology of reproduction in production animals
Long-term impact
The successes achieved by cell therapy in veterinary medicine will serve as preclinical models for applications in human medicine. In addition, tissue engineering using 3D bioprinting will undoubtedly advance in the years to come.
Short-term impact
The minimally invasive approach of Revatis technology promotes the development of animal stem cell bio-banks, which allows the conservation of genetic potential but also deferred clinical uses.
Economic impact
Preventive stem cell banking promotes the use of autologous cell therapy during even minimal trauma or in the early stages of the disease. This promotes faster and better healing.